Commercial Face Masks


Stay Safe
Our non-medical-grade face coverings are made for everyday wear to help keep yourself, your team and your customers safe.
Washable & Reusable
Breathable
Wicks Moisture Away
Designed for both All-day Comfort and Embellishment

Full range of comfy styles designed to meet CDC's facial covering recommendations.
Pocket Mask for Embellishment
Stretch-To-Fit Mask
Adjustable Ear Loop Mask
Wicking Cotton Mask
Youth Masks


HMAS60, P6H0SM
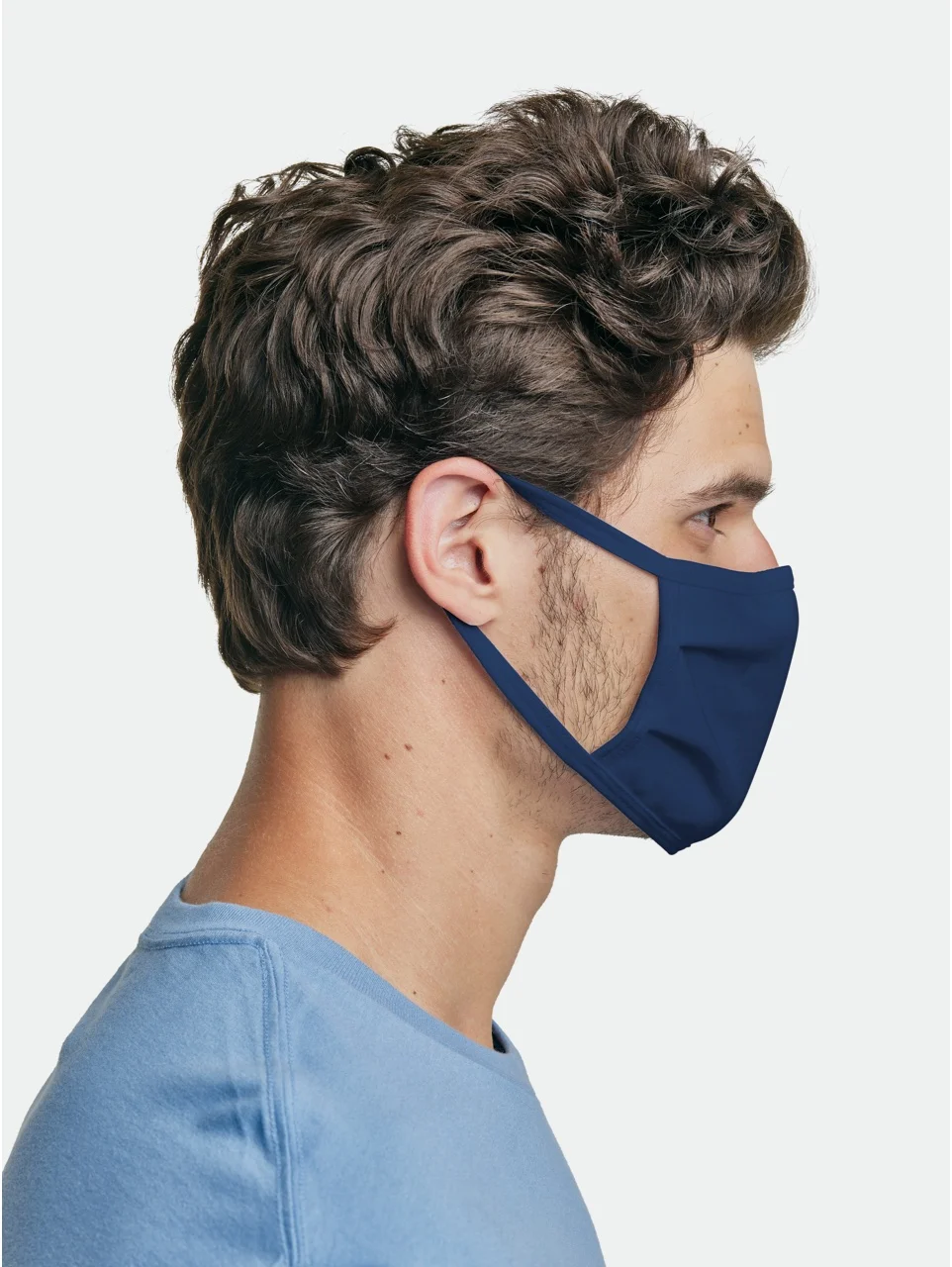
MASKBB

BMSKK5
Looking for Wholesale Face Masks
Hanes Brands B2B is a one-stop mask solution for your school, team, or business.
*Disclaimer
Not recommended for use in a surgical setting or where significant exposure to liquid, bodily or other hazardous fluids may be expected. Shall not be used in a clinical setting where the infection risk level through inhalation exposure is high. Shall not be used in the presence of a high intensity heat source or flammable gas.
MKPKC5 Fabric touching skin: 100% cotton.
MKPKP5 Fabric touching skin: 100% polyester.
BMSKA5 Fabric touching skin: 60% cotton/40% polyester.
BMSKK5 Fabric touching skin: 60% cotton/40% polyester.
MASKBB fabric touching skin: 100% cotton.
This product has not been FDA cleared or approved. This product has been authorized by FDA under an EUA for use by HCP as PPE to help prevent the spread of infection or illness in healthcare settings and by the general public to help slow the spread of the virus during the COVID-19 pandemic.
This product is authorized only for the duration of the declaration that circumstances exist justifying the authorization of the emergency use of medical devices, including alternative products used as medical devices, during the COVID-19 outbreak, under section 564(b)(1) of the Act, 21 U.S.C. Section 360bbb-3(b)(1) unless the authorization is terminated or revoked sooner.
Not authorized for international sale and shipment.
